Renewable Energy
Since its early days, BINA researchers have played a vital role in developing renewable energy applications to address the global challenges of climate change, energy security, and energy poverty, focusing on photovoltaics, energy storage, solar thermal energy, energy conservation, and basic research. BINA's nano-energy experts are world leaders in the techniques forging a path toward practical and green solutions for enhancing people's quality of life by forming a sustainable future.
- Advanced materials
- Solar cells
- Photovoltaics
- Carbon nanotubes
- Ionic liquids
Researchers
-

Prof. Doron Aurbach
972-3-531-8317
We develop the most energetic, high capacity cathodes for Li ion batteries, most suitable for use in electric vehicles
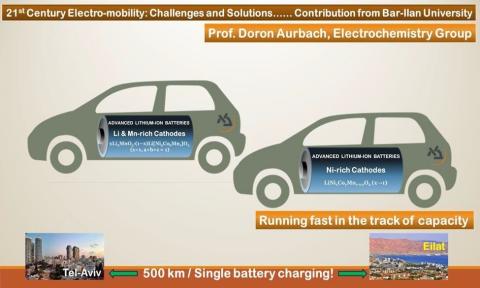
• Li metal based, very high energy density rechargeable batteries.
• Advanced Li ion batteries for electromobility.
• Advance analytical techniques (in-situ observations).
• Batteries for large energy storage (e.g. rechargeable Mg batteries).
• Super-capacitors (very fast energy storage & conversion).
• Water desalination by electrochemical means. -

Dr. Hannah Noa Barad
03-5317876
Multinary Material Systems for Energy and Sustainability
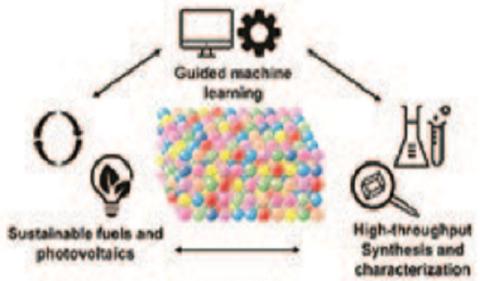
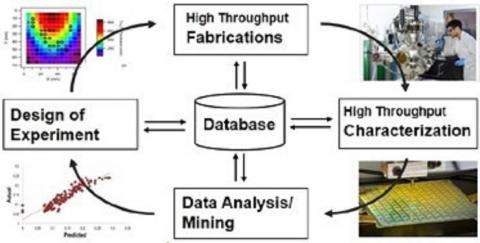
The lab of Hannah-Noa Barad focuses on investigation of multinary (many element) materials that are used for the formation of sustainable fuels and energy. The multinary materials are used as catalysts to form clean sustainable fuels and in photovoltaics. For example, some of the reactions we investigate lead to the formation of H2 (e.g., by water splitting) to be used as an energy source, or carbon-based fuels like CH3CH2OH (e.g., by CO2 reduction). The methods we employ in the lab include machine learning techniques to for rational design and prediction new multinary materials and combinatorial synthesis approaches to fabricate large area libraries of multinary materials with compositional and morphological gradients. The material libraries’ physical, electrical, and chemical properties are then studied using a myriad of different techniques, all of which are built for high-throughput measurements and analysis. All the data we gather in stored in a very large database in the lab. The multinary compounds of interest that we discover are studied in-depth to investigate their working mechanisms and what drives their activity to form clean fuels. The combination of rational design and combinatorial science leads to rapid breakthroughs and state-of-the-art material systems, which will outperform currently used materials and bring about faster and more advanced solutions to the climate crisis.
-

Prof. David Cahen
972-3-531-7876
Alternative Energy
- Photovoltaics (PV), esp. materials for high voltage, low-cost, stable PV
- Combinatorial synthesis and characterization of optoelectronic materials
- Semiconductor materials & device chemistry & physics
- Biomolecular optoelectronics
- Fundamentals of proteins as electronic materials
-

Prof. Moshe Deutsch
972-3-531-8476
Experimental x-ray studies of surface properties of liquid metals
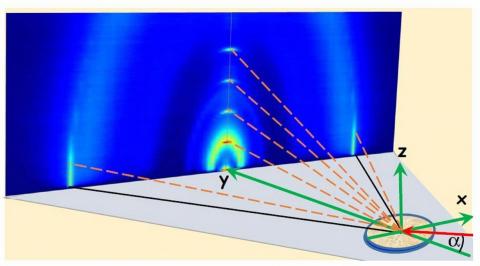
- Experimental x-ray studies of surface properties of liquid metals
- Simple liquids and liquid crystals
- Structure of mono-molecular Langmuir films
- Dynamical x-ray diffraction phenomena in perfect crystals
- High precision crystal binding studies
- X-ray emission and absorption spectroscopy
- Synchrotron radiation and its applications
-

Prof. Lior Elbaz
972-3-738-4252
Fuel Cells and Hydrogen Technologies

• Electrochemistry
• Bio-inspired electrochemistry
• Alternative energy technologies (fuel cells, batteries and photovoltaics)
• Organometallic compounds
• Conductive polymers
• Porphyrins and transition metal complexes
• Ceramic materials
• Semiconductors
• Carbon supports for alternative energy applications (electrodes, electron acceptors) -

Exploring light and matter interactions in materials through quantum electronic structure simulations
Our lab strives to understand and predict how does atomic and electronic structure of materials and molecules effect their optoelectronic and mechanical properties. Understanding microscopic properties of materials, will help in controlling chemical reactions on surfaces and electron transport in nanomaterials, improve optoelectronic properties of devices, and predict novel quantum materials. In the lab we will simulate materials properties, electronic transfer mechanisms, and chemical reactions on the computer. We will develop state of the art electronic
structure for predicting properties of materials, we will focus on nano-materials and 2D quantum materials. -

Prof. Gil Goobes
972-3-5317390
Biomaterials and Advanced Materials group
Research in my group has been focused on revealing and explaining the fundamental interactions that underlie inorganic material formation in nature, a process known as biomineralization. We particularly make use of our expertise in solid-state NMR spectroscopy to analyze the rudimentary processes of biogenic material formation in atomic/molecular level. Unveiling the structure/activity relations in these specialized biomolecules involved in regulation of solid biomaterial formation has been particularly elusive. Using these findings, we develop new biomaterials for hard tissue applications based on rationale guidelines. We implement NMR characterization in materials research to understand interfaces between nanomaterials at great detail and employ molecular insights to design concept materials that are more environment friendly.
-

Prof. Dan Thomas Major
972-3-531-7392 -

Prof. Daniel Nessim
972-3-738-4540
Synthesis of 1D & 2D nanostructures

• Synthesis of carbon nanotubes and understanding of growth mechanisms
• Synthesis of 2D nanocarbons (graphene, GO, rGO)
• Synthesis of 2D metal / sulfidesphosphides - selenides
• Application of synthesized nanostructures to batteries, supercapacitors, fuel cells, heterojunctions, sensors -

Prof. Malachi Noked
972-3-738-4238
Functional thin film for electrochemical devices
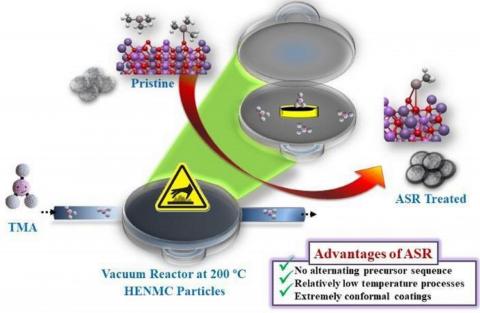
Surface directed chemical reaction in vacuum (atomic/molecular layer deposition ALD/MLD) for:
1. Stabilization of batteries electrodes in implanted medical devices
2. Designing arrays of nano-materials with controlled morphology and structure for electrochemical devices.
3. Modified metallic anode surfaces for next generation rechargeable batteries. -

Prof. Yaakov Tischler
972-3-738-4514
In the Device Spectroscopy Laboratory, we use optical spectroscopy to study nanoscale materials such as molecular organics and more generally nanostructured semiconductors, and then devices composed of these materials
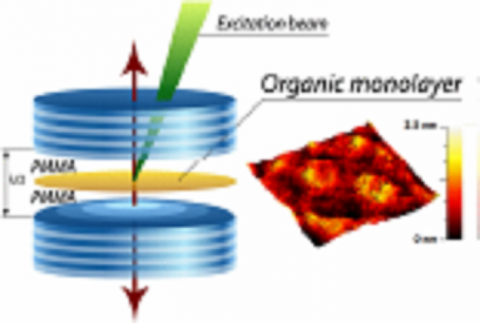
• Coherent coupling in light-matter coupled systems: Organic Lasers, J-aggregates, and Polaritons.
• Ultra-high resolution scanning microcopy and spectroscopy.
• Applications of ultra-fast non-linear spectroscopy for energy sustainability.
• Novel approaches to organic crystal growth and OLED deposition. -

Prof. David Zitoun
972-3-738-4513
The current research focuses on the investigation of the chemical synthesis of materials to promote renewable and green energies
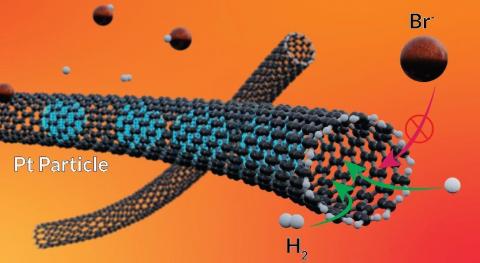
• Photovoltaics and energy storage (batteries and capacitors)
• Inorganic synthesis of semi-conductors, metals and oxides
• Magnetic properties of materials
• Nanostructures: from individual nanoparticles to functional materials




